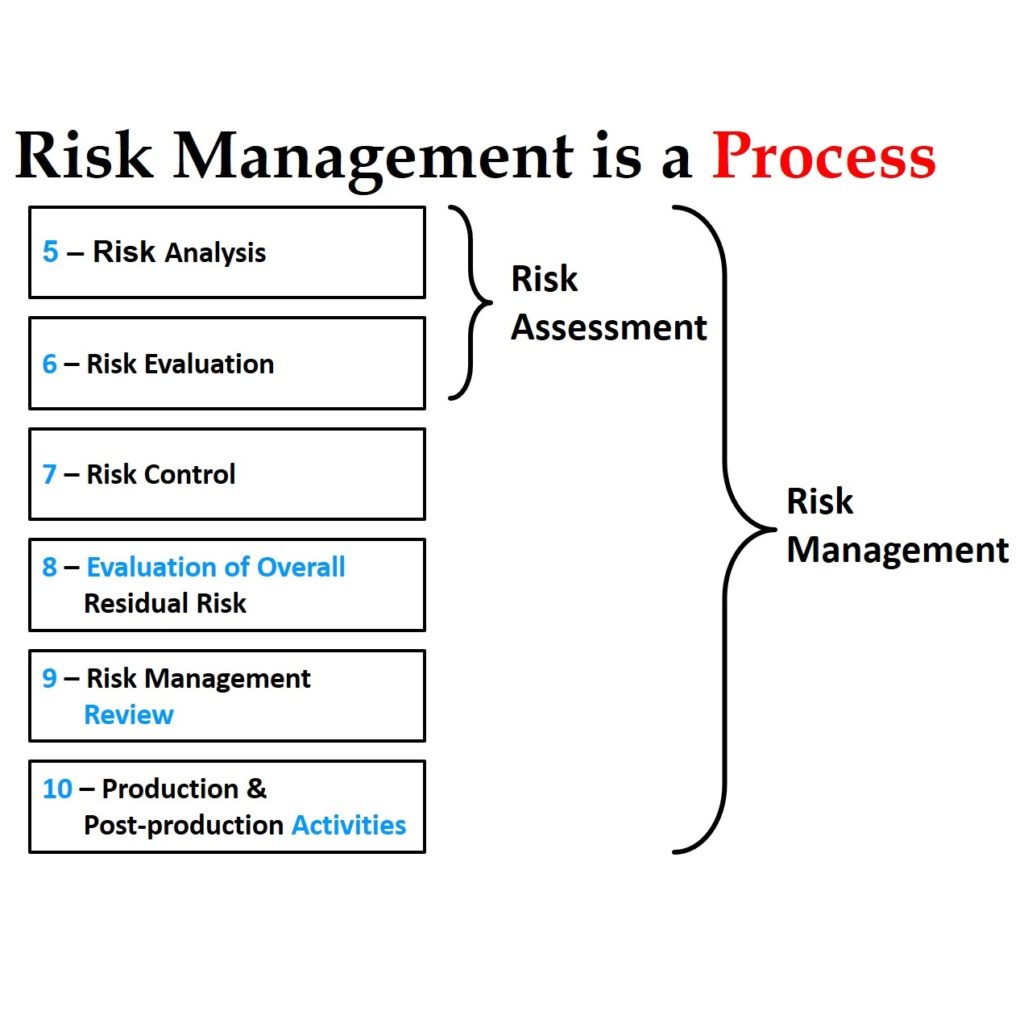
Risk analysis- the process of identifying hazards and calculating the severity of harm with available information. Harm- a physical injury on a person or damage to property or environment.
#Iso 14971 iso#
Some terms defined in the ISO 14971 include: It also provides a risk management review guide and production and post-production activities for medical devices. It also gives some general requirements for a risk management system, It then goes ahead to provide guides on risk analysis, evaluation, and control. ISO 14971 contains definitions of terms that relate to medical device risk management. Previous standards are now used as guides to the current standard. The current state-of-the-art guide was recently republished in 2019. This standard has evolved over time to become a comprehensive guide on risk management for medical devices manufacturers.
#Iso 14971 how to#
Contained in the annexes are in-depth explanations of the guidelines and detailed examples of how to implement the risk management procedures provided. In addition to the guidelines provided in the standard are annexes to the document. Thus a standard framework on the risk management processes for medical devices is important to bring all these stakeholders to agree on the risk management process of the devices. These include manufacturers, doctors, governments, other healthcare practitioners, and patients. Medical devices attract interest from diverse stakeholders. This standard ensures that these risks are monitored from the product design and conception stage, procurement, production, and post-market use. ISO 14971 was developed to provide a standardized process of identifying and monitoring risk across the lifecycle of a medical device. Regulators follow the guidelines provided by this standard in evaluating and allowing medical devices into the market.
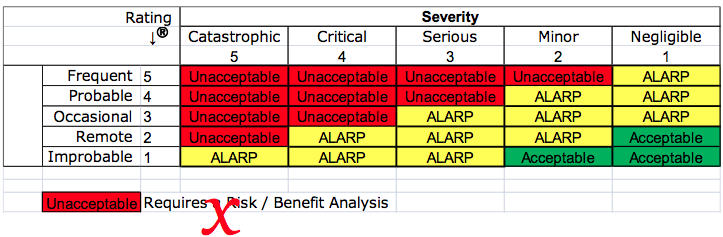
The standard should be used to guide medical device manufacturers in coming up with and following a proper risk management process. ISO 14971 is the current International Standard that provides instructions and guidelines for risk management of medical device manufacturing.

Risk is the probability of occurrence of harm and the extent of the harm if it does occur. Risk management is the process through which a medical device manufacturer can identify and analyze hazards and risks and develop policies to control and monitor the risks. Therefore, as a manufacturer, you should meet all the laid-out risk management standards for your devices to be accepted by regulators and doctors who use them on their customers. When manufacturing medical devices, manufacturers should always consider that the end-user may not have a choice on the device to use. Risk management is an integral part of the manufacture and eventual sale and acceptance of medical devices in the market. Then, you need to make sure that they meet the set regulations and standards for the markets you intend for them to be used. As a medical device manufacturer, the first thing you need to do is to make sure that your devices do what they are supposed to do. Let’s review the background of ISO 14971. ISO 14971 RISK MANAGEMENT FOR MEDICAL DEVICES
